Chapter : 4. Acid Bases & Salts
Basic & Double Salt
लवणों के प्रकार :लवणो के भिन्न प्रकार सामान्य लवण, अम्लीय लवण, क्षारीय लवण तथा द्विकलवण है
3. Basic salt : When a polyacidic base reacts with lesser amount of acid than is necessary for complete neutralization, the salt produced contain hydroxyl group(s) (OH) also. Such a salt is called a basic salt.
EXAMPLES : 1 mole of Pb(OH)2 requires 2 moles of HCl for complete neutralization. But when 1 mole of Pb(OH)2 is made to react with 1 mole of HCl, some Pb(OH)2 is left unreacted. The salt produced is not PbCl2 but Pb(OH)Cl.
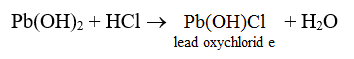
Similarly, when one mole of Bi(OH)3 is reacted with 1 mole of HNO3, the salt Bi(OH)2NO3 is formed.
Bi(OH)3 + HNO3 → Bi(OH)2NO3 + H2O
Salts like Pb(OH)Cl and Bi(OH)2NO3 contain the OH group. These salts are called basic salts, because they can further react with the acids to form H2O and the corresponding normal salts.
Pb(OH)Cl + HCl → PbCl2 + H2O
Bi(OH)2NO3 + HNO3 + Bi(OH)(NO3)2 + H2O
Bi(OH)(NO3)2 + HNO3 → Bi(NO3)3 + H2O
Thus, a basic salt is formed when a poly acidic base reacts with a lesser amount of an acid than is necessary for the formation of a normal salt.
4. Double salt : In a double salt, there are two different negative ions and/or positive ions. For example, the mineral dolomite, CaCO3 • MgCO3 contains both Ca2+and Mg2+ ions. Hence, it is a double salt. Potash alum, K2SO4 • Al2(SO4)3 • 24H2O, also is a double salt.
Double salts exist only in the solid state. When dissolved in water, they break up into a mixture of two separate salts. For example, when potash alum is dissolved in water, it breaks up as follows.

3. Basic salt : When a polyacidic base reacts with lesser amount of acid than is necessary for complete neutralization, the salt produced contain hydroxyl group(s) (OH) also. Such a salt is called a basic salt.
EXAMPLES : 1 mole of Pb(OH)2 requires 2 moles of HCl for complete neutralization. But when 1 mole of Pb(OH)2 is made to react with 1 mole of HCl, some Pb(OH)2 is left unreacted. The salt produced is not PbCl2 but Pb(OH)Cl.
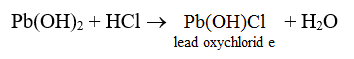
Similarly, when one mole of Bi(OH)3 is reacted with 1 mole of HNO3, the salt Bi(OH)2NO3 is formed.
Bi(OH)3 + HNO3 → Bi(OH)2NO3 + H2O
Salts like Pb(OH)Cl and Bi(OH)2NO3 contain the OH group. These salts are called basic salts, because they can further react with the acids to form H2O and the corresponding normal salts.
Pb(OH)Cl + HCl → PbCl2 + H2O
Bi(OH)2NO3 + HNO3 + Bi(OH)(NO3)2 + H2O
Bi(OH)(NO3)2 + HNO3 → Bi(NO3)3 + H2O
Thus, a basic salt is formed when a poly acidic base reacts with a lesser amount of an acid than is necessary for the formation of a normal salt.
4. Double salt : In a double salt, there are two different negative ions and/or positive ions. For example, the mineral dolomite, CaCO3 • MgCO3 contains both Ca2+and Mg2+ ions. Hence, it is a double salt. Potash alum, K2SO4 • Al2(SO4)3 • 24H2O, also is a double salt.
Double salts exist only in the solid state. When dissolved in water, they break up into a mixture of two separate salts. For example, when potash alum is dissolved in water, it breaks up as follows.

Trending Articles & Blogs
- Physics Tutor, Math Tutor Improve Your Child’s Knowledge
- How to Get Maximum Marks in Examination Preparation Strategy by Dr. Mukesh Shrimali
- 5 Important Tips To Personal Development Apply In Your Daily Life
- Breaking the Barriers Between High School and Higher Education
- 14 Vocational courses after class 12th
- Tips to Get Maximum Marks in Physics Examination
- Get Full Marks in Biology Class 12 CBSE
Download Old Sample Papers For Class X & XII
Download Practical Solutions of Chemistry and Physics for Class 12 with Solutions
Recent Questions Asked
- Newton’s laws of motion asked by Dr. Mukesh Shrimali
- Process of nutrition in Amoeba asked by Rajiv Sharma
- Importance of studying physics subject in school after 10th asked by Rajiv
- Refraction Through Prism in Different Medium asked by Kirti Sharma
- Ratio and Proportion Question asked by Education Desk
- Explain all the 12 tenses with example asked by Qwerty
- Refraction Through Prism in Different Medium asked by Seema Shrimali